Using Safe Equipment for Correct Practice in Ozone Therapy
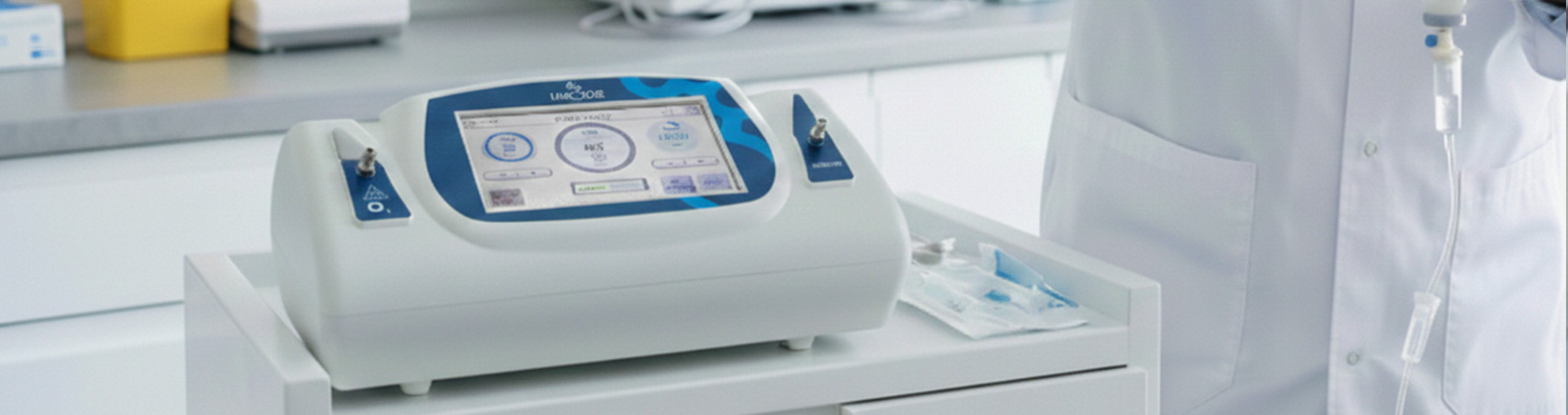
How to Choose a Safe Ozone Therapy Device? A Clinical Guide
Product selection in Ozone Therapy is critical for patient safety.
Learn how to choose the correct, high-quality device with CE Class IIb certification for your clinic with this guide.
Ozone Therapy is increasingly gaining ground in modern medicine due to its benefits, such as strengthening the immune system, accelerating cellular regeneration, and managing chronic pain. However, the success and safety of this powerful treatment depend directly on the quality of the equipment used. Choosing an incorrect or uncertified device not only reduces the efficacy of the treatment but can also pose serious risks to both the patient and the practitioner.
In this guide, we will discuss in detail the critical steps and technical features you should consider when selecting a safe Ozone Therapy device for your clinic.
Devices and Features Used in Ozone Therapy
Various devices and consumables are utilised in Ozone Therapy applications. These include:
- Ozone generators
- Vacuumed glass bottles
- Blood transfusion sets
- Filter sets and other connecting components
Why is Choosing the Correct Equipment Vital?
Ozone (O₃) is a molecule with high oxidative potential. While this property provides its therapeutic effect, it can also cause chemical reactions when it comes into contact with standard medical materials. This situation can create serious risks for patient safety:
- Material Erosion: Plastic materials, blood bags, or metal parts that are not ozone-resistant can erode over time, leading to toxic particles mixing into the treatment.
- Low Treatment Efficacy: The reaction of ozone gas with unsuitable materials causes a decrease in the therapeutic dose that needs to reach the patient.
- Patient Safety Risks: Sets coming into contact with blood must be resistant to medical ozone. Otherwise, undesirable conditions such as haemolysis (the breakdown of red blood cells) may occur.
Checklist for Selecting an Ozone Therapy Device
1. Medical Certification: CE Class IIb is Essential!
Not every ozone generator available on the market is suitable for clinical use.
- CE Class IIb or Class III Certification: This certification verifies that the device is not merely laboratory equipment but is intended for direct medical treatment on humans.
- Risk of Uncertified Devices: Devices without certification are both contrary to regulations and place the clinic under legal liability in case of potential complications.
2. Material Quality and Ozone Resistance
The internal surfaces of the device (such as titanium, quartz glass, and teflon) must be 100% ozone-resistant. Furthermore, the use of materials not specifically manufactured for ozone, such as standard blood and saline bags, must be avoided.
3. Precise Dosage and Concentration Adjustment
Different conditions require different concentrations. It must be ensured that the chosen device can provide precise and consistent dosage adjustments.
Professional Tips for Safe Practice
- Trained Personnel: Application must be performed by experienced healthcare professionals.
- Clinical Protocols: Dosage and the number of sessions should be personalised for each patient.
- Correct Environment: A hygienic and well-ventilated environment is essential.
The Safest Choice for Your Clinic
If you are looking for a reliable solution that meets all these standards, you can explore **Biozon Medical’s** internationally certified products. Elevate the safety of your treatments with our CE Class IIb certified devices.